Atoms, Elements and Compounds
6 Topics | 5 Quizzes
The Periodic Table
10 Topics | 9 Quizzes
Chemical Bonding and Structures
8 Topics | 7 Quizzes
Ionic and Covalent Compounds
8 Topics | 7 Quizzes
Balancing Chemical Equations
5 Topics | 4 Quizzes
Types of Chemical Reactions
7 Topics | 6 Quizzes
Rate of a Chemical Reaction
4 Topics | 3 Quizzes
Factors that Affect Rate of Reaction
7 Topics | 6 Quizzes
Properties of Metals
12 Topics | 11 Quizzes
Metal Reactions
7 Topics | 6 Quizzes
Extraction of Metals
5 Topics | 4 Quizzes
2 | Elements and Compounds

2 | Elements and Compounds
Elements and Compounds
- Pure substances include elements and compounds.
- Elements cannot be broken down into simpler substances.
- Examples of elements include chlorine, sulfur and copper.


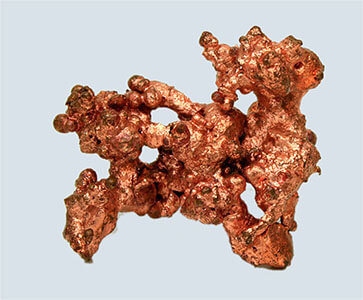
Chlorine, sulfur and copper are elements – they have fixed physical and chemical properties, and cannot be broken down into simpler substances.
(Images: W. Oelen, Wikimedia Commons; Ben Mills, Wikimedia Commons; Jonathan Zander, Wikimedia Commons)
- Compounds can be broken down (chemically) into simpler substances.
- Examples of compounds include water, methane and copper sulfate.

Copper sulfate is a compound – it has fixed physical and chemical properties, but can be broken down into simpler substances (the elements copper, sulfur and oxygen).
(Image: Stephanb, Wikimedia Commons)
